Details of the Drug
General Information of Drug (ID: DM2HU9I)
| Drug Name |
Triamterene
|
||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Ademin; Ademine; Amteren; Anjal; Dazid; Diarol; Dinazide; Diren; Ditak; Diucelpin; Diurene; Diutensat; Diuteren; Dyberzide; Dyren; Dyrenium; Dytac; Dytenzide; Esiteren; Hidiurese; Hydrene; Hypertorr; Isobar; Jatropur; Jenateren; Kalspare; Masuharmin; Nephral; Noridil; Noridyl; Pterofen; Pterophene; Renezide; Reviten; Taturil; Teriam; Teridin; Triamizide; Triampur; Triamteren; Triamterena; Triamterenum; Triamteril; Triamterine; Triamthiazid; Tricilone; Trispan; Triteren; Triurene; Trizid; Turfa; Uretren; Urocaudal; Goldshield Brand of Triamterene; Jorba Brand of Triamterene; Pter ophene; SmithKline Beecham Brand of Triamterene; TRIAMTERENE USP; Thiazid Wolff; Triamteril complex; Wellspring Brand of Triamterene; Fluss 40; SKF 8542; T 4143; Ademin(e); Apo-triazide; Dyrenium (TN); Pteridine deriv. 11; SALI-PUREN; SK&F 8542; SK-8542; Tri-Span; Triamterena [INN-Spanish]; Triamterenum [INN-Latin]; SK&F-8542; Triamterene (JP15/USP/INN); Triamterene [USAN:INN:BAN:JAN]; 2,4,7-Triamino-6-fenilpteridina; 2,4,7-Triamino-6-fenilpteridina [Italian]; 2,4,7-Triamino-6-phenylpteridine; 6-PHENYL-2,4,7-TRIAMINO PTERIDINE; 6-Phenyl-2,4,7-pteridinetriamine; 6-Phenyl-2,4,7-triaminopteridine; 6-phenylpteridine-2,4,7-triamine
|
||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Diuretics
|
||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||
| Structure |
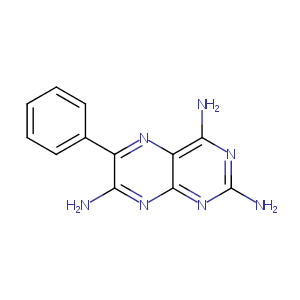 |
||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 253.26 | |||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1 | ||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 1 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 7 | ||||||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Transporter (DTP) |
|
|||||||||||||||||||||||||||||||||||||||||
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Triamterene
Coadministration of a Drug Treating the Disease Different from Triamterene (Comorbidity)
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4329). | ||||
|---|---|---|---|---|---|
| 2 | Triamterene FDA Label | ||||
| 3 | DailyMed Label: DYRENIUM (triamterene) oral capsules | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Gilfrich HJ, Kremer G, Mohrke W, Mutschler E, Volger KD: Pharmacokinetics of triamterene after i.v. administration to man: determination of bioavailability. Eur J Clin Pharmacol. 1983;25(2):237-41. | ||||
| 6 | PRO-TRIAZIDE (Triamterene 50 mg and Hydrochlorothiazide 25 mg) - Product Monograph | ||||
| 7 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 8 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 9 | Effects of the serine/threonine kinase SGK1 on the epithelial Na(+) channel (ENaC) and CFTR: implications for cystic fibrosis. Cell Physiol Biochem. 2001;11(4):209-18. | ||||
| 10 | Identification of novel substrates and structure-activity relationship of cellular uptake mediated by human organic cation transporters 1 and 2. J Med Chem. 2013 Sep 26;56(18):7232-42. | ||||
| 11 | Rate-limiting biotransformation of triamterene is mediated by CYP1A2. Int J Clin Pharmacol Ther. 2005 Jul;43(7):327-34. | ||||
| 12 | Identification of environmental chemicals that activate p53 signaling after in vitro metabolic activation. Arch Toxicol. 2022 Jul;96(7):1975-1987. doi: 10.1007/s00204-022-03291-5. Epub 2022 Apr 18. | ||||
| 13 | Population-based in vitro hazard and concentration-response assessment of chemicals: the 1000 genomes high-throughput screening study. Environ Health Perspect. 2015 May;123(5):458-66. doi: 10.1289/ehp.1408775. Epub 2015 Jan 13. | ||||
| 14 | Identification of chemical compounds that induce HIF-1alpha activity. Toxicol Sci. 2009 Nov;112(1):153-63. | ||||
| 15 | Product Information. Aldactone (spironolactone). Searle, Skokie, IL. | ||||
| 16 | Marcy TR, Ripley TL "Aldosterone antagonists in the treatment of heart failure." Am J Health Syst Pharm 63 (2006): 49-58. [PMID: 16373465] | ||||
| 17 | Dean S, Kendall MJ, Potter S, Thompson MH, Jackson DA "Nadolol in combination with indapamide and xipamide in resistant hypertensives." Eur J Clin Pharmacol 28 (1985): 29-33. [PMID: 3987783] | ||||
| 18 | Brown DD, Dormois JC, Abraham GN, et al "Effect of furosemide on the renal excretion of digoxin." Clin Pharmacol Ther 20 (1976): 395-400. [PMID: 975715] | ||||
| 19 | Aronowitz JS, Chakos MH, Safferman AZ, Lieberman JA "Syncope associated with the combination of clozapine and enalapril." J Clin Psychopharmacol 14 (1994): 429-30. [PMID: 7884028] | ||||
| 20 | Ban TA "Drug interactions with psychoactive drugs." Dis Nerv Syst 36 (1975): 164-6. [PMID: 1116424] | ||||
| 21 | Agencia Espaola de Medicamentos y Productos Sanitarios Healthcare "Centro de informacion online de medicamentos de la AEMPS - CIMA.". | ||||
| 22 | Muller FO, Schall R, Devaal AC, Groenewoud G, Hundt HKL, Middle MV "Influence of meloxicam on furosemide pharmacokinetics and pharmacodynamics in healthy volunteers." Eur J Clin Pharmacol 48 (1995): 247-51. [PMID: 7589049] | ||||
| 23 | Product Information. Savella (milnacipran). Forest Pharmaceuticals, St. Louis, MO. | ||||
| 24 | Warrington SJ, Ankier SI, Turner P "Evaluation of possible interactions between ethanol and trazodone or amitriptyline." Neuropsychobiology 15 (1986): 31-7. [PMID: 3725002] | ||||
| 25 | Product Information. Yasmin (drospirenone-ethinyl estradiol) Berlex Laboratories, Richmond, CA. | ||||
| 26 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 27 | Yassa R, Nastase C, Camille Y, Henderson M, Belzile L, Beland F "Carbamazepine, diuretics, and hyponatremia: a possible interaction." J Clin Psychiatry 48 (1987): 281-3. [PMID: 3597330] | ||||
| 28 | Product Information. Aptiom (eslicarbazepine). Sunovion Pharmaceuticals Inc, Marlborough, MA. | ||||
| 29 | Muirhead MR, Somogyi AA, Rolan PE, Bochner F "Effect of cimetidine on renal and hepatic drug elimination: studies with triamterene." Clin Pharmacol Ther 40 (1986): 400-7. [PMID: 3757403] | ||||
| 30 | Product Information. Midamor (amiloride). Merck & Co, Inc, West Point, PA. | ||||
| 31 | Product Information. Tekturna (aliskiren). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 32 | Jarman PR, Mather HM "Diabetes may be independent risk factor for hyperkalaemia." BMJ 327 (2003): 812. [PMID: 14525902] | ||||
| 33 | Jarman PR, Kehely AM, Mather HM "Hyperkalaemia in diabetes: prevalence and associations." Postgrad Med J 71 (1995): 551-2. [PMID: 7479468] | ||||
| 34 | McNay JL, Oran E "Possible predisposition of diabetic patients to hyperkalemia following administration of potassium-retaining diuretic, amiloride (MK 870)." Metabolism 19 (1970): 58-70. [PMID: 5410663] | ||||
| 35 | Product Information. Samsca (tolvaptan). Otsuka American Pharmaceuticals Inc, Rockville, MD. | ||||
| 36 | Blakely KM, Drucker AM, Rosen CF "Drug-induced photosensitivity-an update: Culprit drugs, prevention and management." Drug Saf 42 (2019): 827-47. [PMID: 30888626] | ||||
| 37 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 38 | Perazella MA "Drug-induced hyperkalemia: old culprits and new offenders." Am J Med 109 (2000): 307-14. [PMID: 10996582] | ||||
| 39 | Canaday DH, Johnson JR "Hyperkalemia in elderly patients receiving standard doses of trimethoprim-sulfamethoxazole." Ann Intern Med 120 (1994): 438. [PMID: 8304666] | ||||
| 40 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
